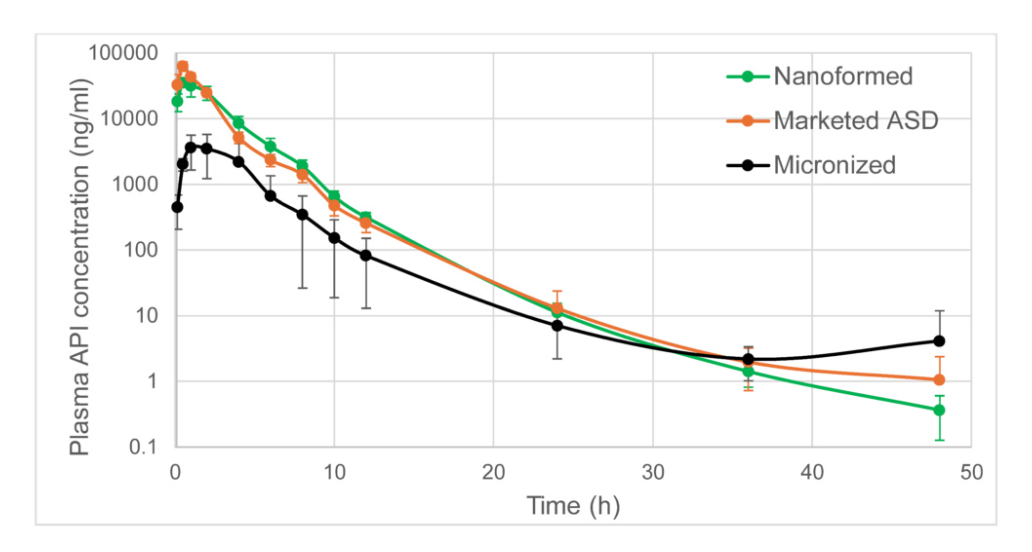
We can NanoImprove your Medicine:
Nanoform Biologics
Nanoform Biologics can deliver your large molecule products in more stable powdered forms, as high concentration injections, or for inhaled delivery.
LATEST PRESS RELEASES & NEWS
To read more of our latest news and see our recent articles and videos, visit our media centre
Subscribe to hear the latest news about
Loading...