Technologies and Services

Our nanoforming technologies, formulation and analytical services span the full range of drug development from small-molecule to large-molecule therapeutics. We accelerate your time to clinic while also increasing possibilities and probabilities of success in taking your product to market by developing the optimal nanoformulation.
STARMAP®
Our STARMAP® online platform leverages cutting-edge sparse-data AI to pick winners among candidate molecules that are predicted to be best amenable to CESS®-powered nanoforming and that exhibit optimal production characteristics.
Case studies
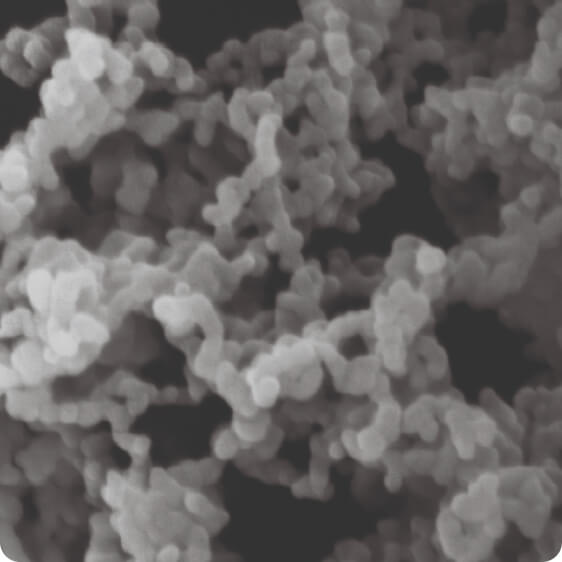
Our multi-patented Controlled Expansion of Supercritical Solutions (CESS®) technology can enable the creation of uniform small-molecule API nanoparticles as small as 10 nm directly from solution.

Analytical Capabilities
Analytical chemistry plays a crucial role in characterizing and understanding materials produced through nanoforming.

IP Protection
Pharmaceutical companies can maximize their return on investment by ensuring their products have a strong intellectual property framework.

Nanoforming Science
Nanoform’s Controlled Expansion of Supercritical Solutions (CESS®) technology is uniquely capable of producing pure API particles as small as 10 nm.